LIPOSUCTION TEXTBOOK
The Tumescent Technique By Jeffrey A. Klein MD
LIPOSUCTION TEXTBOOK
Chapter 23:
Tumescent Formulations
No standard, official, or rigidly prescribed formulation exists for tumescent anesthetic solutions. Concentrations of the lidocaine and epinephrine should depend on the areas treated and the clinical situation.
In this book the word dosage implies the total amount of a drug given relative to the patient’s weight in kilograms (mg/kg); a dose is a quantity of a medicine given at one time and measured in milligrams (mg).
Tumescent Caveats
Before discussing the formulation of tumescent solutions for local anesthesia, the following concepts and warnings must be emphasized to minimize the risks of lidocaine toxicity.
- Understand maximum safe dosage. The maximum recommended dosage of lidocaine is 45 mg/kg in relatively thin patients and 50 mg/kg in obese patients (see Chapter 21). I avoid using more than 55 mg/kg. At doses less than 55 mg/kg, plasma lidocaine concentrations greater than 6 μg/ml may result from adverse drug interactions mediated by the inhibition of cytochrome P450 3A4 (CYP3A4). Patients occasionally experience nausea and vomiting approximately 12 hours after tumescent infiltration of 50 to 55 mg/kg; in such patients, measured plasma lidocaine concentration has never exceeded 3.5 μg/ml. To minimize this risk, it is preferable not to exceed 45 mg/kg of tumescent lidocaine. These dosage limitations assume no drug interactions and no unimpaired function of hepatic CYP3A4 (see Chapters 18 and 21).
- Use signed written orders. The surgeon must provide explicit written orders for the formulation of the tumescent local anesthetic solution. Standard policy should be that no solutions are mixed unless the surgeon has signed the orders and the orders are in the patient’s chart. All orders for tumescent anesthesia should include documentation of the patient’s weight (kg), maximum desired dosage (mg/kg), and exact amount of each drug to be included in the tumescent solution expressed in milligrams per liter (mg/L) or milliequivalents per liter (mEq/L).
- Know dosage given. The surgeon, anesthesiologist, and nurses must always know the patient’s final total milligram dose (mg) and the milligram per kilogram dosage (mg/kg). For each patient, the surgeon’s orders must also explicitly state the surgeon’s determination of the maximum safe dosage of tumescent lidocaine in mg/kg. Surgeons have been charged with criminal negligence (but not convicted) because of apparent errors in lidocaine dosing.
- Specify dosage in terms of milligrams. The orders for formulating the solution of local anesthesia for the tumescent technique should specify the exact total milligrams of lidocaine and epinephrine and the milliequivalents of sodium bicarbonate per liter of tumescent solvent (mg/L and mEq/L). The orders should not be given in terms of milliliters of 1% lidocaine per liter of solution [(ml/L)(10 mg/ml)]; to do so increases the risk of inadvertent dosing errors. It is much easier to determine the mg/kg dose of lidocaine when the concentration of lidocaine in each liter is specified in mg/L.
- Use only 1% lidocaine. Preferably, a surgeon’s formulary should stock only commercial vials of 1% lidocaine. The risk of a inadvertent overdose is vastly increased when vials of 2% lidocaine are available. Patients have received double the intended dose of lidocaine when 2% lidocaine was used instead of the intended 1% solution. Although these cases did not result in serious toxicity, each incident of incorrect dosage represents a potential disaster. Epidural anesthesia may require 2% lidocaine, but no dermatologic surgical procedures require more than 1% lidocaine.
- Ensure licensed medical personnel prepare solution. Only well-trained personnel should mix the tumescent solution. Surgical operating room (OR) technicians or medical assistants are usually not licensed to prepare or administer drugs and anesthetics. Unlicensed personnel are more likely to make errors in the interpretation of anesthetic orders or the actual mixture of the tumescent anesthetic solution. Mixing the tumescent local anesthetic solution requires “eyes-on” or hands-on supervision of licensed medical personnel.
- Have solution prepared at surgery. To avoid medication errors, the tumescent anesthetic solution should be prepared in the OR at the time of surgery. Preparing the tumescent anesthetic solution in large batches and far in advance may increase the risk of unrecognized contamination or inadvertent dosage errors. Concerns about safety outweigh the possible convenience of preparing tumescent solutions for multiple patients.
- Save all empty bottles. All empty vials of lidocaine and epinephrine should be temporarily saved until the surgical procedure is completed. This precautionary strategy allows personnel to double-check the total lidocaine or epinephrine dosage. If a discrepancy exists between an intended dosage and the number of empty vials of lidocaine or epinephrine, all the remaining tumescent anesthetic mixtures must be discarded and new mixtures prepared.
- Avoid postoperative sedatives. Use of diazepam (Valium) 24 hours after tumescent liposuction is relatively contraindicated. Diazepam or other sedatives may increase the risk of lidocaine toxicity by inhibiting CYP3A4 or by impairing ventilation and producing respiratory acidosis.
- Use normal saline as preferred tumescent solvent. As discussed later, normal saline (NS), also known as physiologic saline or 0.9% sodium chloride (NaCl), is the preferred solvent for dilute solutions of local anesthetics using the tumescent technique. When safe and reasonable volumes of tumescent solution are infiltrated, no significant difference exists between NS and lactated Ringer’s solution (LR).
- Ensure anesthesiologist and surgeon share responsibility. All physicians, surgeons, and anesthesiologists in the OR are responsible for a surgical patient’s safety. When providing systemic anesthesia, the anesthesiologist must be completely informed about and concur with the total dosage of tumescent lidocaine. An anesthesiologist who is not cognizant of the lidocaine dosage (mg/kg) or the volume of subcutaneously infiltrated tumescent fluid may be unable to avoid adverse drug interactions or systemic fluid overload.
Anesthesiologists must be familiar with the pharmacology and pathophysiology of tumescent liposuction to be in compliance with American Society of Anesthesiologists (ASA) standards. For regional anesthesia, the ASA states that “anesthesiologists should assume responsibility for all aspects of anesthesia care,” including outpatient anesthesia.1
Safety Warning
Safe doses of tumescent (very dilute) lidocaine and epinephrine are not the same for “out-of-the-bottle” commercial (considerably more concentrated) lidocaine. Whereas the safe maximum dosage of tumescent lidocaine (with epinephrine) at concentrations of 0.05% to 0.15% is 45 to 50 mg/kg, the traditional dosage limitation for commercial lidocaine (with epinephrine) at concentrations of 0.5%, 1%, or 2% remains valid at 7 mg/kg. All physicians should be extremely careful to recognize this vital distinction.
Lidocaine and Epinephrine Concentrations
The concentration of lidocaine and epinephrine in an anesthetic solution should vary according to the clinical requirements. No “correct” or sanctioned concentration of lidocaine or epinephrine exists for tumescent local anesthesia.
This chapter’s recommendations for concentration of tumescent lidocaine with liposuction of various body areas have been developed empirically. Years of experience and careful observation have helped define an estimate of the optimal concentrations. The goal is to determine the minimal concentration for each component of the anesthetic solution that consistently permits painless liposuction. Areas that are especially fibrous, such as the upper abdomen, breast, and back, also tend to be associated with increased surgical bleeding. The more fibrous areas tend to require higher concentrations of lidocaine and epinephrine. Less fibrous and less sensitive areas require lower concentrations. Recommended concentrations are simply guidelines and are always subject to modification (Table 23-1).
Use of smaller cannulas is associated with less discomfort and a smaller probability of encountering an area of painful liposuction. Thus smaller cannulas allow the use of lower drug concentrations. Surgical technique that initiates liposuction using the smallest cannulas and then increases cannula size sequentially causes less discomfort than beginning liposuction with relatively large cannulas.
If several areas are treated by liposuction on the same day, the surgeon may need to use the lowest concentrations so as not to exceed the maximum safe dosage limits (mg/kg).
Epinephrine
Epinephrine, a hormone derived from the adrenal medulla, is also known as adrenaline. Pharmacologically it acts as both an alpha and a beta agonist, increasing heart rate as well as peripheral vasoconstriction and blood pressure. More importantly for tumescent anesthesia, epinephrine is a potent capillary vasoconstrictor responsible for the dramatic hemostasis and the slow systemic lidocaine absorption associated with the tumescent technique.
Tachycardia. Patients may give a history of some type of adverse reaction to epinephrine. Typically this involved dental anesthesia in which the patient experienced the unpleasant pharmacologic effects of rapid systemic absorption. Injection of a drug into the highly vascular oral (periodontal, gingival, or buccal) mucosa is more likely to produce rapid systemic absorption than injection into less vascular tissue. Rapid absorption is a pharmacologic phenomenon, not an allergic reaction.
Rapid absorption of epinephrine can produce tachycardia, tremors, and anxiety. In my experience, however, patients who have experienced tachycardia from rapid absorption of epinephrine after dental anesthesia do not have a similar reaction with tumescent anesthesia for liposuction.
Liposuction surgeons should be cautious in assessing patients with a confusing history of an adverse reaction to epinephrine. Patients taking pseudoephedrine for nasal decongestion or “health food” supplements that contain ephedrine-like chemicals are predisposed to epinephrine-associated tachycardia. Also, the patient may have an undiagnosed primary cardiac arrhythmia (dysrhythmia), an occult cardiac valvular disease with intermittent tachycardia, or a subclinical metabolic disorder (e.g., hyperthyroidism, carcinoid, pheochromocytoma). When in doubt, the surgeon should consider an internal medicine consultation.
If the patient’s history is clearly consistent with rapid absorption of epinephrine and the consequent pharmacologic response with tachycardia, tumescent liposuction probably poses minimal risk. In some patients, especially older persons, it is wise initially to limit the amount of liposuction to relatively small volumes. Once the first procedure has been completed without evidence of tachycardia, tremor, or an anxiety reaction, the surgeon can proceed 1 or 2 months later with a standard dose of epinephrine for tumescent anesthesia.
The routine use of clonidine (0.1 mg) given preoperatively to patients without bradycardia or hypotension has greatly reduced the incidence of intraoperative and postoperative tachycardia with tumescent local anesthesia.
Regional Variation. The physician can vary the concentration of epinephrine depending on the particular area that is being targeted for tumescent liposuction. In areas that tend to be associated with increased intraoperative bleeding, such as upper abdomen, back and flank, and especially fibrous areas of fat, it is reasonable to use 1 mg of epinephrine/L tumescent anesthetic solution. For other areas, 0.65 mg of epinephrine/L is usually sufficient.
Safety Checks
Basic Solution. A basic, minimally effective solution of tumescent local anesthesia consists of 500 mg of lidocaine, 0.5 mg of epinephrine, and 10 mEq of sodium bicarbonate in 1 L of NS. It is specifically intended for the surgeon who wants to check the completeness of the anesthesia just before beginning liposuction. This may be necessary if someone other than the surgeon, such as a registered nurse or another physician, has done the infiltration.
Checking the completeness of the local anesthesia is intended to detect areas of subcutaneous fat that are not completely anesthetized. This should be done immediately before initiating liposuction. While advancing an infiltrating cannula throughout the tumescent compartment of fat, the surgeon or anesthetist should instruct the patient to indicate any area of incomplete anesthesia by saying the word “there” whenever the cannula causes even a minimally painful sensation. The surgeon can then infiltrate additional anesthetic solution exactly where it is needed.
The minimal concentration of lidocaine in the basic solution is usually sufficient to achieve complete anesthesia and vasoconstriction.
Complete Records. The surgeon must insist that the staff assiduously maintain complete, legible records that document the total milligram dose and concentration of local anesthetic ingredients in each liter of tumescent solution. The important ingredients include lidocaine, epinephrine, and sodium bicarbonate.
Traditionally, physicians have only used relatively small doses of subcutaneous local anesthetics. Few medical students or surgical residents have ever been trained to document or record the exact amount of local anesthetic used. Specific preoperative orders for subcutaneous infiltration of local anesthesia are rarely written. Even now, with increased use of the relatively high doses of lidocaine employed with tumescent anesthesia, many surgeons still have a casual, nonrigorous approach to documentation of local anesthesia dosages.
Poor records are dangerous, below the standard of care, and often associated with malpractice. Careless anesthesia records combined with a patient death may result in a prosecution for criminal negligence. In two such cases the surgeon’s notes did not accurately document the total dose of lidocaine; both patients died after receiving general anesthesia (inhalational or propofol) plus tumescent local anesthesia, which was not well documented. Although both cases resulted in charges of criminal negligence, neither resulted in a conviction. Criminal negligence is not covered by malpractice insurance, and a conviction can involve time in prison.
Shelf Life of Solution. Manufacturers of 0.9% NaCl and LR state, “When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.”
It is safer to prepare the tumescent anesthetic solution on the day of surgery in the OR. Medicolegal considerations favor the use of anesthetic solutions that are freshly prepared in the OR immediately before surgery. It would be difficult to defend the use of a “stale” solution if a complication were attributable to pharmacologic instability or bacterial contamination.
An error in the preparation of the tumescent anesthetic solution is more easily detected if the anesthetic is mixed immediately before or during the surgery. By saving the empty bottles of lidocaine and empty vials of epinephrine, the total dosage of these drugs can be double-checked simply by counting the empty containers. If tumescent anesthetic solution is prepared a day or more in advance or outside of the OR, an inadvertent overdose of lidocaine in one or more bags of saline might not be detected.
The efficacy of a tumescent anesthetic solution is not maximal when mixed several days before surgery. The shelf life varies as a function of pH, temperature, and concentrations of other solutes. In particular, the vasoconstrictive properties of epinephrine are not stable at a pH of 5 or greater. There is a greater risk that lidocaine might precipitate from an older solution or from a solution with a higher pH. If a liter bag of tumescent anesthetic solution has been prepared days in advance, the physician cannot always be certain that it has been properly stored.
Sodium Bicarbonate and Bupivacaine. Bupivacaine is a larger, less water-soluble molecule than lidocaine. Adding 5 mEq of sodium bicarbonate to 50 ml of bupivacaine (0.75%) will result in the immediate precipitation of the bupivacaine. Injecting such a suspension intradermally or subcutaneously has caused full-thickness dermal necrosis.
Therefore the physician should never add sodium bicarbonate to bupivacaine.
Adding 10 mEq/L of sodium bicarbonate to a dilute solution of tumescent lidocaine does not cause the lidocaine to precipitate. It is prudent not to exceed this amount to minimize the risk of lidocaine precipitation.
Triamcinolone. The addition of triamcinolone (10 mg/L) once was considered beneficial in reducing the incidence of focal postliposuction subcutaneous inflammation, or panniculitis.2 In my early experience with tumescent liposuction, approximately 2% to 4% of patients had focally tender, pink, warm, sterile subcutaneous nodules that did not respond to antibiotics. When postliposuction panniculitis was treated with a course of oral antibiotics and prednisone (10 mg/day), however, the condition improved dramatically within 24 to 48 hours.
It was concluded that including triamcinolone in the anesthetic solution might prevent postliposuction panniculitis. In fact, triamcinolone seemed to reduce the incidence of this focal inflammation.
Careful clinical study, however, showed that the decreased panniculitis resulted from the use of open drainage and bimodal compression. The closure of incisions with suture was eliminated about the same time as triamcinolone was introduced. The simple act of encouraging the rapid drainage of residual tumescent fluid and the inflammatory postsurgical subcutaneous exudate was the true cause for improved healing.
The only advantage of including triamcinolone in the tumescent anesthetic solution is a slight reduction in postoperative soreness, which is apparent 3 to 5 days after surgery. This slight effect is clinically insignificant. On the other hand, the surgeon must be concerned about the possible increased risk of postliposuction infection that might result from steroid-induced impairment of immune function. Thus, it seems prudent to avoid unnecessary use of triamcinolone.
Tumescent Solvents and Fluid Homeostasis
The choice of solvent, into which the lidocaine and epinephrine are added, is an important aspect of the tumescent technique. The solvent should be isotonic and the pH nearly physiologic. With conservative liposuction, no significant difference probably exists between NS and LR in terms of safety or efficacy. With excessively large-volume liposuction procedures, however, the differences in formulation may be clinically relevant.
First, no studies detail the pharmacokinetics of parenteral (subcutaneous infiltration or intravenous [IV] infusion) administration of more than 6 L of NS or LR in the setting of tumescent liposuction. Most “expert” opinions on fluid and electrolyte homeostasis in liposuction are substantiated only by anecdotal experience or clinical dogma.
Tumescent infiltration of 5 L of solution was closely studied in one patient on two occasions.2 No liposuction was done after the first infiltration. Two weeks later the second infiltration was followed by liposuction of 1.5 L of supranatant fat. No IV fluids were given during either procedure. The published results showed that, with or without liposuction, urine specific gravity decreased and urine output was greater than 70 ml/hr, indicating no IV fluid deficit (see Figure 19-2, D and E). Thus IV fluids were not necessary for tumescent liposuction of 1.5 L of supranatant fat.
In my experience, liposuction of up to 4 L of supranatant fat in moderately obese females does not require IV fluids. This assumes that patients are fully alert and can take oral fluids whenever they are thirsty.
Normal Saline
The most common lidocaine solvent for the tumescent technique is isotonic physiologic saline (0.9% NaCl), or NS. As defined by the United States Pharmacopeia (USP), NS contains 154 mEq/L of both sodium and chloride (Table 23-2). Plasma contains 142 mEq Na/L.
Typically, sodium bicarbonate (NaHCO3, 10 mEq/L) is added to the lidocaine solution to neutralize the pH and thus reduce the stinging pain that otherwise occurs when acidic commercial lidocaine is infiltrated subcutaneously in an alert patient. Thus a liter of tumescent solvent will contain 164 mEq of sodium.
Ringer’s Solution Without Lactate
Ringer’s solution, without lactate, was developed by Sidney Ringer (1834-1910), a professor of medicine at the University of London and one of the first scientific clinical pharmacologists.3 Ringer is best known for his research on the effects of blood electrolytes on cardiac function.4-7
When an assistant mistakenly injected tap water instead of distilled water into a frog heart, Ringer noticed that tap water did not produce the immediate cardiac arrest expected with distilled water. He tied a string around a frog heart and studied the effects of bathing the heart in different solutions, using a tonometer to measure the force of contractions. He showed that a 0.75% NaCl solution profoundly impaired contractions and that adding dried bovine blood to the solution restored normal contractions. Similarly, adding minute doses of potassium salts had the same beneficial effect on cardiac contraction as adding dried bovine blood. Ringer also found that adding NaHCO3 to the saline solution had no beneficial effect on cardiac function.
Ringer concluded that “saline solution, to which is added one ten-thousandth part of potassium chloride, makes an excellent circulating fluid in experiments with the detached heart.” The results of Ringer’s research were not appreciated for many years.
Other Solvents and Potassium. One application of Ringer’s experiments might be in autologous fat transplantation. Ringer’s solution might be the preferred solvent for tumescent anesthetic before harvesting donor fat for transplanting into another part of the body. Because of the large quantity of endogenous potassium ion in the extracellular fluid of a healthy human, however, using Ringer’s solution or NS as a tumescent solvent would be clinically the same. Furthermore, in vitro potassium requirements may not be the same for heart muscle cells and other cells.
Ringer’s solution, LR, and NS are similar when used as solvents for tumescent anesthesia. The dogma that LR is “more physiologic” than NS is only applicable to the patient with severe hypokalemia. Healthy liposuction patients undergoing a safe amount of liposuction have no potassium deficit. The cell lysis associated with liposuction may even increase the local potassium concentration as a result of spilling intracellular potassium into the interstitial space.
Hartmann’s Solution (Lactated Ringer’s Solution)
Alexis Frank Hartmann (1898-1964), an American clinical biochemist and pediatrician at Washington University in St. Louis, studied diabetes and diabetic ketoacidosis in children, which led to a search for an optimal isotonic alkalizing solution. He is responsible for putting lactate in Ringer’s solution.
Sodium bicarbonate is less than ideal for the treatment of acidosis in children. At therapeutic doses, bicarbonate is highly irritating when given by IV infusion. Solutions of bicarbonate are relatively unstable and cannot be sterilized simply by boiling. If bicarbonate corrects the acidosis too rapidly, it can precipitate a profound and dangerous metabolic alkalosis. Hartmann’s research was directed toward finding an alternative to NaHCO3. The treatment of ketoacidosis required an isotonic solution with a moderate alkalizing effect.
Hartmann and a colleague determined that a one-sixth molar solution of sodium lactate is (1) isotonic and can be sterilized by heat without decomposition, (2) metabolized in the liver, (3) safe in patients with renal or hepatic insufficiency, (4) potentially equivalent to 172 mEq of NaHCO3, and (5) equivalent to 600 ml of 5% dextrose in antiketogenic effect.8-11 Lactate is used instead of bicarbonate because it is more stable in IV fluids during storage.12
Lactate Metabolism. The lactate in LR is not instantaneously converted to bicarbonate after infusion. Two metabolic pathways, gluconeogenesis and oxidation, are responsible for the conversion of lactate to bicarbonate. Approximately 70% of lactate is metabolized by gluconeogenesis, which is initiated by the enzyme pyruvate kinase, principally in the liver but also in the kidneys. Oxidation accounts for 30% of the lactate metabolism.
The overall equation for the metabolism of lactate by gluconeogenesis is as follows:
2CH3CHOHCOO– + 2H+ ⇌ C6H12O6
(Lactate) (Glucose)
The following is the overall equation for the metabolism of lactate by oxidation:
2CH3CHOHCOO– + 2H+ + 3O2 → 3H2O + 3CO2
With a half-life of approximately 15 minutes in a healthy subject, the complete metabolic conversion of lactate to bicarbonate requires 1 to 2 hours.
Sodium Bicarbonate. A tumescent anesthetic solution without sodium bicarbonate (10 mEq NaHCO3/L) is needlessly painful on infiltration in an awake patient. Adding sufficient NaHCO3 neutralizes the pH of an acidic solution of local anesthetic. When LR or NS is the solvent for a tumescent solution of lidocaine and epinephrine, the addition of NaHCO3 is necessary for painless infiltration.
Complications. LR (USP) contains 130 mEq/L of sodium, 109 mEq/L of chloride, 29 mEq/L of lactate, 4 mEq/L of potassium, and 2.7 mEq/L of calcium.
The average adult produces 1200 to 1500 mmol of lactate/day, or approximately 50 to 60 mmol/hr.13 The liver metabolizes about 60% of this, and the kidney metabolizes or excretes the remaining 40%. The liver and kidney can accommodate large lactate infusions without immediate detriment. Large doses of LR containing 29 mEq/L of lactate produce a delayed iatrogenic metabolic alkalosis.
LR has been associated with perioperative complications. Hemodilution associated with LR during surgery may predispose to deep venous thrombosis.14
The administration of sodium lactate in LR can cause alkalosis as well as exacerbate preexisting alkalosis. Lactate is metabolized in the liver faster than the kidneys can excrete sodium. The resulting anion deficiency is compensated by an increased production of , and alkalosis occurs.
Alkalosis may cause cardiac dysrhythmias.15 Alkalosis reduces the effects of vagal stimulation and reduces the level of bradycardia that might result from any given stimulus. Metabolic alkalosis increases oxygen requirements and exacerbates cardiac dysrhythmias caused by hypoxia or hypokalemia. In alkalotic patients, NS should be used instead of LR for the same indications.
Nerve Compression. Acute median nerve compression has been described in three patients as the result of significant IV infusion of LR in tumescent liposuction.16 Each surgical procedure was completed in less than an hour. The patients received a total of 6100 ml, 9000 ml, and 7500 ml of LR, respectively, of which only 2000 to 2500 ml was infiltrated subcutaneously.
From a pharmacokinetic perspective, both LR and NS behave as a drug, for which the rate of absorption, distribution, and elimination can be described in standard mathematic terminology. Systemic absorption of tumescent solvent is insignificant for the first 6 hours after infiltration. Thus all the systemic effects of fluid overload can be attributed to the excessive IV fluid infusion. The systemic fluid overload, as manifested by acute median nerve paralysis, can be avoided by not using IV fluids with tumescent liposuction.
Acidosis and Alkalosis. As noted, some surgeons prefer to use LR as the solvent for their tumescent lidocaine solutions because of LR’s “sodium-sparing effect” or because LR is “more physiologic” than NS. Surgical textbooks provide little information to substantiate this preference.17
The lactate in LR is principally intended to produce a gradual reduction of metabolic acidosis. In healthy patients with no acidosis, the excess bicarbonate produces a transient mild metabolic alkalosis, which is promptly corrected by renal bicarbonate excretion. Critically ill patients, however, may be obligate excreters of acidic urine and be unable to excrete the bicarbonate load associated with LR, which in turn may cause metabolic alkalosis and produce renal potassium loss.18
Because the body contains a huge supply of intracellular potassium as well as the potassium in the extracellular fluid, the K+ in LR is unnecessary, and physiologic saline (NS) is sufficient for the clinical replacement of intravascular fluids with crystalloid solutions.
Transfusion with blood products becomes necessary long before any possible risk of potassium deficit from using 0.9% NaCl instead of LR.
Lactate as Inept Buffer System. A buffered solution resists a change in pH when either hydroxide (OH–) ions or protons (H+) are added.19 Blood must be a very efficient buffered solution. Despite the constant metabolic production of acids and bases, the pH of blood must be maintained within a narrow range necessary for cellular viability. Lactic acid, a metabolic product of anaerobic metabolism in muscle, is a weak acid with Ka of 1.4 × 10–4 and pKa of 3.85.
The pKa of a weak acid used in a buffered solution should be as close as possible to the desired pH of the solution. Because a pKa of 3.85 is far from the pH of 6.2 of LR and from the pH of 7.4 of plasma, the lactate in LR is not an effective buffer.
Basic chemistry provides the following:
HC3H5O3 ⇌ H+ +
and
Ka = ([H+][])/[HC3H5O3] = 1.43 10–4
where [] = 29 mmol/L = 2.9 × 10–2 mol/L, which is the standard concentration of lactate ions in LR. Because the pH of commercial LR is approximately 6.2, it follows that [H1] = 10–6.2 = 6.3 × 10–7 mol/L. Rearranging the expression for Ka results in the following:
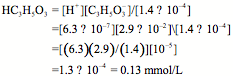
Thus the ratio [HA]/[A–] = [HC3H5O3]/[] = [0.13 mmol/L]/[29 mmol/L] = 0.13/29 = 4.5 × 10–3, which is far from the ideal buffer, where [HA]/[A–] = 1.
LR has no significant chemical buffering action. LR provides only an indirect source of physiologically active bicarbonate, after lactate has been metabolized by the liver. It is a fallacy to use LR assuming that it provides any significant buffering action. A solution of tumescent lidocaine with epinephrine in LR is acidic, unless it has been neutralized by the addition of sodium bicarbonate. When the tumescent solvent is LR, the anesthetic solution is too acidic and produces significant stinging pain on infiltration of tumescent lidocaine; in contrast, when 10 mEq/L of sodium bicarbonate is added to neutralize the pH of the LR, the pain is virtually eliminated.
Cellular Distribution
The water in the body is subdivided into different compartments, including intracellular fluid (ICF) and extracellular fluid (ECF). The ECF consists mainly of the intravascular fluid (plasma), or interstitial fluid.
The distribution of water between various compartments within the body is determined by the relative concentration of discrete solute particles, regardless of size or electrical charge. The number of solute particles per unit volume is described in terms of mOsm/L (osmolarity) or mOsm/kg H2O (osmolality).
In clinical medicine, solute particles in body fluids are best quantified of mOsm according to osmolarity (e.g., mOsm/L of plasma). In physiology, osmolality is preferred because it is invariant under changes of temperature and independent of the volume occupied by the solutes in the solution. The osmolality of plasma is 290 mOsm/kg H2O.
The total body water is 36 L (60% of body weight), with the ECF and ICF containing 12 and 24 L, respectively.
Chloride Load. Rapid expansion of ECF volume with fluids that do not contain bicarbonate () temporarily reduces concentration in the ECF as a result of excessive Cl–anions.20 For example, a rapid infusion of NS produces relatively mild hyperchloremia, which causes a compensatory shift of extracellular into the intracellular space. This type of normal-anion-gap acidosis is mild and rapidly corrected by respiratory compensation and by the kidneys, which excrete and Cl–.
The kidneys filter 4000 mmol of per day, most of which is resorbed, with 80% in the proximal tubules and 20% in the distal tubules. Because NS is not buffered and does not contain a net excess of H+ ions, this mild transient hyperchloremic acidosis does not produce systemic acidosis.
Sodium Load. Sodium ions are excluded from the intracellular space by the Na+-K–transmembrane pump. An excessive amount of total body sodium produces an osmotic pressure gradient that pulls water across cell membranes and increases ECF volume. Also, ingested free water is retained in the ECF by the extra sodium.
In progressive degrees, ECF overload is manifested by peripheral dependent interstitial edema, then by intravascular fluid overload with incipient congestive heart failure, and in the extreme, by pulmonary edema.
Based on this observation, some surgeons propose using LR with its lower sodium content (130 mEq/L) instead of NS (154 mEq/L Na) as the solvent for tumescent lidocaine solutions. The use of LR is of no benefit except for tumescent infiltration of greater than 10 to 12 L of anesthetic solution.
Sodium Chloride and Molarity. The molecular weight of NaCl is 58.5 g/mol and that of Na is 22.9 g/mol. By definition, one mole of a substance contains Avogadro’s number of units. The weight of one mole of NaCl is its molecular weight.
The molarity of a solute is defined as the number of moles of a solute per liter of solvent. A liter of water is approximately 1 kg, depending on the water’s temperature. NS consists of a 0.9% solution of NaCl (0.9 g NaCl/100 ml = 9 g NaCl/L). Thus the molarity of NS is as follows:
(9 g NaCl/L H2O)(mol NaCl/58.5 g NaCl)
= 0.154 mol NaCl/L
= 154 mmol NaCl/L
= 154 mEq Na+/L
Thus 1 L of tumescent solvent (NS plus 10 mEq Na/L in the form of NaHCO3) contains approximately 164 mEq Na1/L.
Saline and Lactate. A liter of NS with NaHCO3 and a liter of LR differ in sodium content by 34 mEq/L (164 –130 mEq/L). This difference is clinically insignificant for routine tumescent infiltration of 5 to 7 L or less of subcutaneous fluid. The time required for systemic absorption of this volume of tumescent fluid is approximately 24 to 36 hours.
However, 5 L of tumescent solvent (NS with 10 mEq NaHCO3) exceeds the amount of sodium in 5 L of LR by 170 mEq Na+ = 170 mmol NaCl = 0.17 mol NaCl = (0.17 mol) (58.5 g NaCl/mol) = 9.95 g NaCl. To put this in perspective, a 1-pound (450-mg) bag of pretzels contains approximately 9 g of sodium chloride.
Thus LR does have some sodium-sparing effect, but again, this is only significant in the range of dangerously excessive tumescent liposuction that infiltrates more than 10 to 12 L of solution.
The difference between NS and LR should be of little concern for a surgeon who is primarily concerned with patient safety. Problems with fluid overload are easily avoided by not attempting huge-volume liposuction in a single surgical procedure. A large volume of aspirated fat removed by serial liposuctions is much safer than a single surgery.
Intravenous Fluids
Burn Analogy
The pathophysiology of liposuction has been compared to that of major thermal trauma. This burn analogy, however, does not hold for the pathophysiology of tumescent liposuction. The fluid resuscitation requirements for tumescent liposuction and burns are distinctly different. Resuscitation after a major burn requires large volumes of IV fluids. In contrast, with tumescent liposuction, IV fluids are unnecessary and contraindicated because of the serious risk of systemic fluid overload.
Severe burns are associated with coagulation necrosis, altered capillary permeability, and large-volume extravasation of plasma into burned tissues. Hypoproteinemia induces edema in remote nonburned tissue, as well as significant intravascular fluid depletion. Because LR has 130 mEq/L of sodium, it is hypotonic with respect to sodium and provides some free water. Surgical textbooks recommend LR as the resuscitation fluid of choice for major burns.21
In contrast, tumescent liposuction requires no IV fluids. Tumescent infiltration superhydrates the targeted tissues, and the systemic absorption of tumescent fluid prevents any significant intravascular fluid deficit. Tumescent liposuction produces a net flux of isotonic fluid into the vascular space rather than the intravascular fluid deficit seen with a large burn.
With respect to intravascular fluid homeostasis, tumescent liposuction is more similar to a transurethral resection of the prostate (TURP) than to a burn. To extend the analogy further, with the tumescent technique, as with the TURP syndrome or the operative hysteroscopy intravascular absorption (OHIA) syndrome, the surgeon must guard against intravascular overload and hyponatremia.22
Excessive Liposuction and Hemodilution
If the volumes of aspirated fat and parenteral fluid administration are so large that sodium overloading is a concern, too much liposuction is being done. Huge volumes of parenteral isotonic fluid and excessive sodium are rarely a concern when tumescent liposuction is limited to safe volumes of supranatant fat (less than 3% to 5% of body weight).
In a healthy patient the maximum safe volume of tumescent infiltration is limited by hemodilution, not by sodium overload. By the time a patient without congestive heart failure has received enough NS to cause concern about sodium overload, the deleterious effects of hypercoagulability associated with hemodilution will have supervened.
Surgeons who perform huge-volume liposuction (supranatant fat volume 5% of body mass or greater) are often compelled to administer IV fluids. IV fluid supplementation in the setting of tumescent liposuction greatly increases the risks of liposuction. Excessive intravascular absorption of LR with simultaneous IV fluid infusion can produce pulmonary edema and a hemodilution-induced hypercoagulable state. Hemodilution with LR use during surgery may predispose to deep venous thrombosis.14
With or without liposuction, 5 L of tumescent solution decreases the hematocrit by 10%; this is probably a nonlinear effect. Even greater volumes of tumescent solution may produce an increasing degree of hemodilution, perhaps exponentially.
With tumescent liposuction, IV fluids produce additional hemodilution. Excessive hemodilution predisposes to massive generalized postoperative edema, as well as hypercoagulability and perhaps disseminated intravascular coagulation (DIC). Liposuction under general anesthesia, using 15 L of a subcutaneous tumescent solution plus 18 L of IV LR, has resulted in fatal DIC (see Case Report 14-1).
No persuasive physiologic or pharmacologic rationale exists for using either LR or NS as the solvent for tumescent anesthesia. Massive doses of isotonic solutions should be avoided, however, as should megaliposuction. Supplemental IV infusion in conjunction with tumescent infiltration is an absolute contraindication.
References
- American Society of Anesthesiologists: Standards, guidelines and statements, 1998, The Society.
- Klein JA: Tumescent technique for local anesthesia improves safety in large-volume liposuction, Plast Reconstr Surg 92: 1085-1098, 1993.
- Lee JA: Sydney Ringer (1834-1910) and Alexis Hartmann (1898-1964), Anaesthesia 36:115-121, 1981.
- Ringer S: Regarding the action of the hydrate of soda, hydrate of ammonia, and the hydrate of potash on the ventricle of the frog’s heart, J Physiol (Lond) 3:195-202, 1880/1882.
- Ringer S: Concerning the influence exerted by each of the constituents of the blood on the contraction of the ventricle, J Physiol (Lond) 3:389-393, 1880/1882.
- Ringer S: A further contribution regarding the influence of the different constituents of the blood on the contraction of the ventricle, J Physiol (Lond) 4:29-42, 1883/1884.
- Ringer S: A third contribution regarding the influence of the inorganic constituents of the blood on ventricular contraction, J Physiol (Lond) 4:222-225, 1883/1884.
- White SA, Goldhill DR: Is Hartmann’s the solution? Anaesthesia 52:422-427, 1997.
- Hartmann AF, Senn MJE: Studies in the metabolism of sodium r-lactate. 1. Response of normal human subjects to the intravenous injection of sodium r-lactate, J Clin Invest 11:327-336, 1932.
- Hartmann AF, Senn MJE: Studies in the metabolism of sodium r-lactate. 2. Response of human subjects with acidosis to the intravenous injection of sodium r-lactate, J Clin Invest 11:337-344, 1932.
- Hartmann AF, Senn MJE: Studies in the metabolism of sodium r-lactate. 3. Response of human subjects with liver damage, disturbed mineral and water balance, and renal insufficiency to the intravenous injection of sodium r-lactate, J Clin Invest 11:345-355, 1932.
- Shires GT III, Barber A, Shires GT: Fluid and electrolyte management of the surgical patient. In Schwartz SI, Shires GT, Spencer FC, et al: Principles of surgery, ed 7, New York, 1999, McGraw-Hill.
- Woerlee GM: Common perioperative problems and the anaesthetist, Boston, 1988, Kluwer Academic Press.
- Janvrin SB, Davies G, Greenhalgh RM: Postoperative deep vein thrombosis caused by intravenous fluids during surgery, Br J Surg 67:690-693, 1980.
- Vaughan-Williams EM, Whyte JM: Chemosensitivity of cardiac muscle, J Physiol 189:119-137, 1967.
- Lombardi AS, Quirke TE, Rauscher G: Acute median nerve compression associated with tumescent fluid administration, Plast Reconstr Surg 102:235-237, 1998.
- Schwartz SI, Shires GT, Spenser FC, et al: Principles of surgery, ed 7, New York, 1999, McGraw-Hill.
- Cohen RD, Simpson R: Lactate metabolism, Anesthesiology 43:661-673, 1975.
- Zumdahl SS: Chemistry, ed 3, Lexington, Mass, 1993, Heath.
- Winters RW et al: The mechanism of acidosis produced by hyperosmotic infusions, J Clin Invest 43:647, 1964.
- Goodwin CW, Finkelstein JL, Madden MR: Burns. In Schwartz SI, Shires GT, Spencer FC, editors: Principles of surgery, ed 6, New York, 1994, McGraw-Hill.
- Jackson S, Lampe G: Operative hysteroscopy intravascular absorption syndrome, West J Med 162:53-54, 1995.
| TABLE 23-1 Recommended Concentrations for Effective Tumescent Anesthesia for Liposuction | |||
| Areas | Lidocaine (mg/L) | Epinephrine (mg/L) | Sodium Bicarbonate (mEq/L) |
| Basic/checking | 500 | 0.5 | 10 |
| Hips; lateral, medial, and anterior thighs; knees | 700-750 | 0.65 | 10 |
| Back; male flanks; arms | 1000 | 0.65-1.0 | 10 |
| Female abdomen (medial) | 1000-1250 | 1.0 | 10 |
| Male abdomen (medial) and male breasts | 1250 | 1.0 | 10 |
| Abdomen (lateral) | 750 | 0.65 | 10 |
| Female breasts; chin, cheek, and jowls | 1500 | 1.5 | 10 |
| Facial resurfacing (CO2 laser) | 600 mg/250 ml | 1 mg/250 ml | 5 mEq/250 ml |
| TABLE 23-2 Electrolyte Content (mmol/L) of Anesthetic Solvents and Extracellular Fluid | ||||||
| Solution/Fluid | Na+ Ions | Cl– Ions | K+ Ions | Ca++ Ions | Lactate Ions | Osmolality (mOsm/L) |
| Normal saline (NS), USP | 154 | 154 | 308 | |||
| Normal saline (NS), BP | 150 | 150 | ||||
| Lactated Ringer’s solution, USP | 130 | 109 | 4 | 1.4 | 29 | 273.4 |
| Hartmann’s solution, BP | 131 | 111 | 5 | 2 | 29 | |
| Extracellular fluid (ECF) | 142 | 103 | 4 | 2.5 | 290* | |
USP, United States Pharmacopeia; BP, British Pharmacopoeia.
*Plasma osmolality is largely the sum of osmolalities from plasma electrolytes, glucose, and urea.
